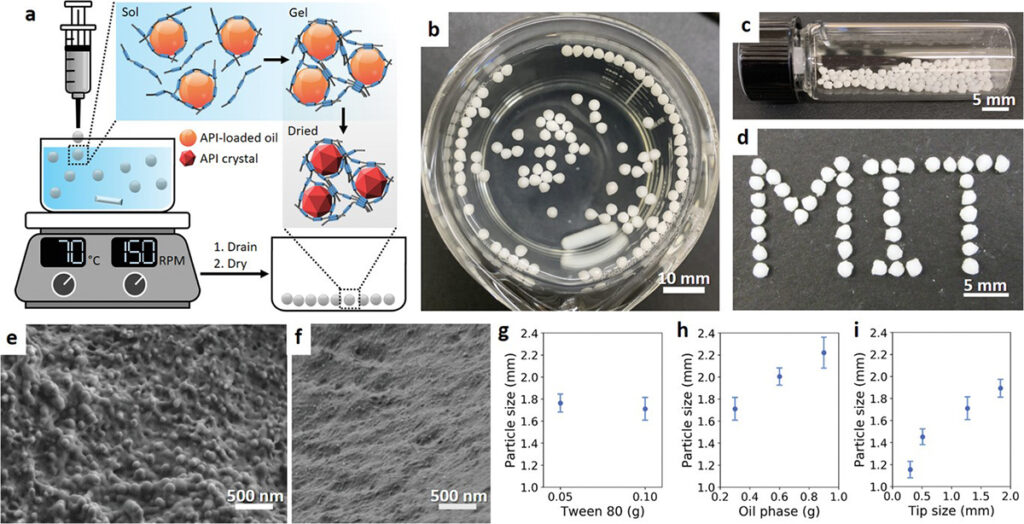
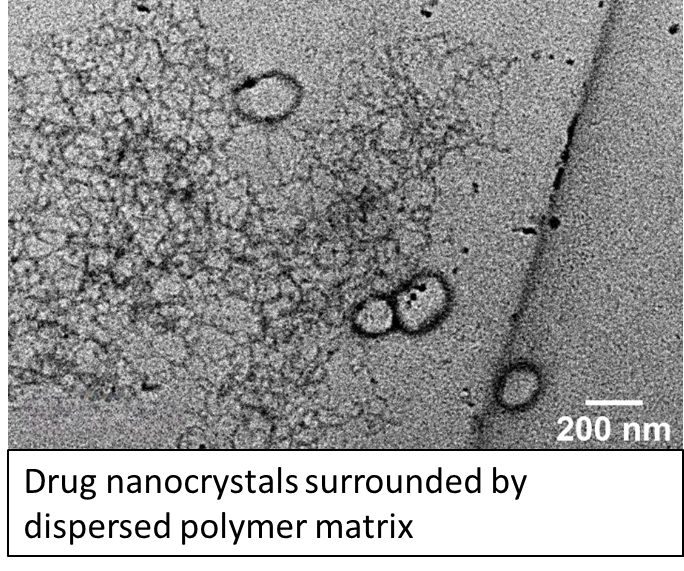
Hydrophobic active pharmaceutical ingredients are ubiquitous in the drug development pipelines, but their poor bioavailability often prevents their translation into the clinic. This poor bioavailability is attributed to the slow dissolution kinetics of hydrophobic APIs in the aqueous environment of the gastrointestinal (GI) tract. Conventional formulation processes for small-molecule therapeutics rely on ‘top-down’ approaches, like media milling, to reduce crystal size and improve dissolution kinetics, but these processes are difficult to optimize and do not enable the flexibility to design customizable drug release profiles for a given drug’s pharmacokinetics. Our lab has developed many technologies to engineer ‘bottom-up’ technologies that produce drug nanocrystals embedded in polymer matrices using nanodomains as templates for crystallization. These drug-polymer co-processing technologies can enable flexible and modular formulation and allows for multi-scale control over the properties of composite particles. This research area is a collaborative effort with colleagues in Professor Doyle’s group at the Singapore MIT Alliance for Research and Technology (SMART). Ongoing efforts include extending these technologies to formulate complex composite hydrogel particles as wells as understanding how mechanistic molecular interactions can inform intelligent formulation design.
References:
1. Chen, L.-H. & Doyle, P. S. Design and Use of a Thermogelling Methylcellulose Nanoemulsion to Formulate Nanocrystalline Oral Dosage Forms. Adv. Mater. 33, e2008618 (2021).
2. Chen, L.-H., Cheng, L.-C. & Doyle, P. S. Nanoemulsion-Loaded Capsules for Controlled Delivery of Lipophilic Active Ingredients. Adv. Sci. 7, 2001677 (2020).
3. Badruddoza, A. Z. M., Gupta, A., Myerson, A. S., Trout, B. L. & Doyle, P. S. Low energy nanoemulsions as templates for the formulation of hydrophobic drugs. Adv. Ther. 1, 1700020 (2018).
4. Ng, D. Z. L. et al. Control of Drug-Excipient Particle Attributes with Droplet Microfluidic-based Extractive Solidification Enables Improved Powder Rheology. Pharm. Res. 39, 411–421 (2022).